TARGETING CANCER’S MOST commonly mutated pathway
Why RAS/MAPK?
There are approximately five million new cases of cancer per year with RAS/MAPK pathway alterations, most of which have limited or no treatment options. While the RAS/MAPK pathway has been well characterized and validated based on the development and approval of multiple compounds targeting discrete signaling nodes in the pathway, most of these compounds face resistance and tolerability challenges, highlighting the need for new approaches to target this important signaling cascade. The RAS/MAPK pathway is implicated in approximately one-third of all solid tumors, including colorectal cancer (CRC), non-small cell lung cancer (NSCLC), and pancreatic ductal adenocarcinoma (PDAC).
MAPKlamp & More
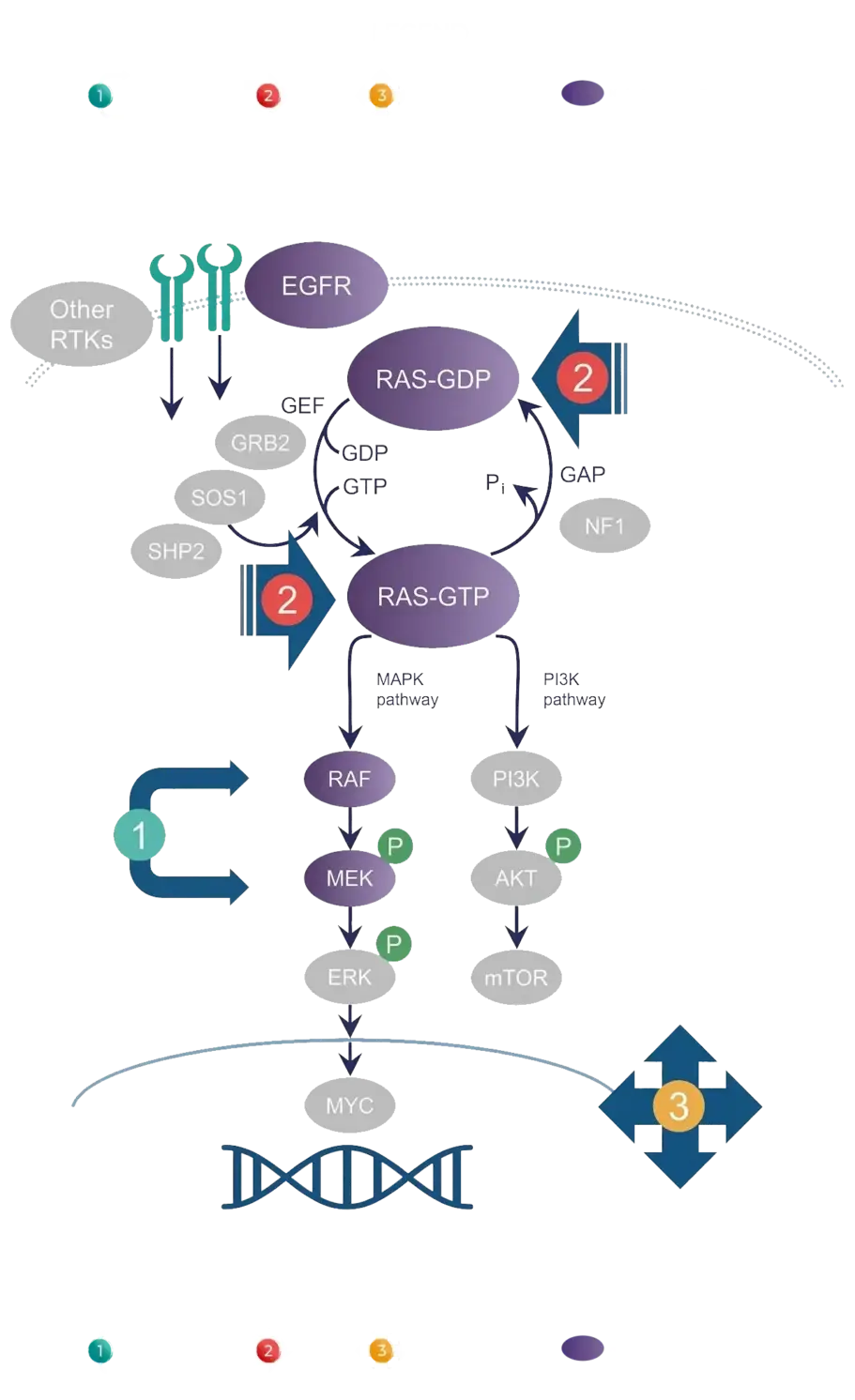
Using innovative science and working with world-class collaborators, we are taking a holistic, modality-agnostic approach to effectively shut down the RAS/MAPK pathway. We are not just targeting individual signaling nodes. Rather, we seek to turn off multiple nodes and cooperative mechanisms along the pathway in parallel. To accomplish this, we are pursuing three therapeutic strategies that work together to comprehensively, and perhaps synergistically, shut down the RAS/MAPK pathway.
Our 3 therapeutic strategies
To learn more about our three therapeutic strategies and how they can help us overcome RAS/MAPK pathway-driven cancers, click on the numbers below.
Target upstream and downstream MAPK nodes
with single agents and
clamp oncogenic drivers (MAPKlamp™) with combinations
Target RAS directly
with single agents and combinations with
upstream, downstream,
and escape route targeted
therapies
Target escape routes
enabled by other proteins
or pathways to further
disrupt RAS/MAPK
pathway signaling
Our strategic focus on the RAS/MAPK pathway allows us to comprehensively target every critical node in the pathway that could drive signaling. In fact, we currently have programs targeting each of the nodes colored in purple below.
Presentations
ERAS-0015
ERAS-4001
DISCOVERY
Publications
RAS-GDP and RAS-GTP
- K-Ras(G12C) inhibitors allosterically control GTP affinity and effector interactionsNature, November 2013
Jonathan M. Ostrem, Ulf Peters, Martin L. Sos, et al. - Targeting KRAS DirectlyAnnual Review of Cancer Biology, March 2018
Frank McCormick - Ras Binder Induces a Modified Switch-II Pocket in GTP and GDP StatesCell Chemical Biology, December 2017
Daniel R. Gentile, Manoj K. Rathinaswamy, Meredith L. Jenkins, et al. - RAS Proteins and Their Regulators in Human DiseaseCell, June 2017
Dhirendra K. Simanshu, Dwight V. Nissley, Frank McCormick, et al.